Over 3,000 bidimensional materials are now in the Materials Cloud database
By Nicola Nosengo, NCCR MARVEL
The Materials Cloud, the online resource for computational materials science created by NCCR MARVEL, includes a database of 2D materials that were obtained computationally by going through a list of experimentally known 3D materials and looking for those that, because of their electronic properties, look like they could be easily exfoliated to create bidimensional materials.
The collection has just been greatly enriched thanks to a refinement of the computational protocol and an extension of the 3D materials used as source, and now counts over 3,000 candidate 2D structures.
The latest extension is described in an article published in ACS Nano, of which former MARVEL member Davide Campi, previously with Nicola Marzari’s group at EPFL and now at the University of Milan-Bicocca, is the first author.
Campi and his MARVEL coauthors (Nicolas Mounet, Marco Gibertini, Giovanni Pizzi, and Nicola Marzari) adopted a slightly revised version of a computational method that had already been described in an earlier article in Nature Nanotechnology, and that uses high-throughput, van-der-Waals density-functional theory calculations to find materials that can be easily exfoliated from existing layered, tridimensional structures. “The only thing that we changed is about a parameter that we use to differentiate weak van-der-Waals bonds and strong covalent bonds” Campi explains. “In this new work we allowed it to have a wider range of values. On one side this leads to more false positives that we then had to exclude with further calculations, but on the other side it lets us capture some cases that we had missed in the previous analysis. We noticed that a few structures had been excluded because they appeared as being 3D, even though we had experimental evidence that they had been synthesized in 2D. We decided to have a bit more computational cost overall, in order to be sure not to miss any material”.
In addition, the team expanded the list of 3D materials from which to source. They went back to the Inorganic Crystal Structure Database (ICSD) and the Crystallography Open Database (COD) that had been used previously and that in the meantime have been updated. And they added a third database, the Pauling File (MPDS). Overall, they considered 9,306 layered candidates, 3689 of which were not included in the previous screening. To narrow down the list, they first optimized the structures found in the 3D database by taking them to their theoretical equilibrium volume. This allows to exclude structures that appeared as layered based on geometrical screening, but no longer do so once relaxed – either because the original structure had errors, or because they were obtained under extreme pressure conditions. “We aim at zero pressure, the goal is to have a material that can be exfoliated at atmospheric pressure” explains Campi.
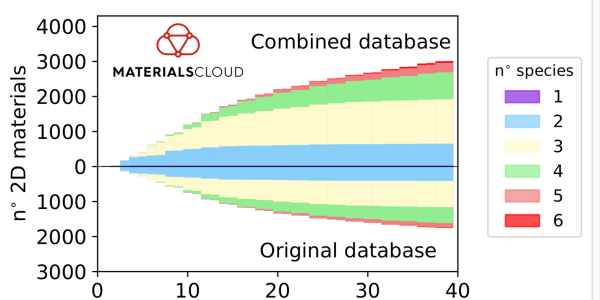
Number of structures as a function of the number of atoms in the primitive cell (top, exfoliable 2D structures in the overall database; bottom, exfoliable 2D structures obtained previously).
Another type of geometrical screening allows a further selection of candidate materials. “On those that still appear layered, we take all the components that have weak bonds, and we simulate their isolation in the void,” says Campi. This allows to compare the energy of these systems in the void with the energy of the 3D structure, which is normally lower. This comparison gives the material’s binding energy. The smaller it is, the easier it should be to peel the material and turn it into a 2D material.
The result was a list of 1252 new monolayers, bringing the total now present in the Materials Cloud to 3,077 structures.
The researchers also grouped these materials in prototypes, groups of materials that share the same fundamental arrangement of atoms in the 2D structure. “For many materials, the atoms change but the structure of their disposition in space is the same” says Campi. “To define the, we ignored the chemical element as well as the absolute position in space, considering only the re-scaled, relative positions of the elements with respect to each other”. On one side, this can give ideas to create new materials, he explains. “If we find holes in these classes because a material was never synthesized, but it seems reasonable to replace an atom with another, for example because they belong to the same group in the periodic table, we may want to try and make it”. On the other side, researchers can get new ideas by studying how electrical properties change from one prototype to the other.
The article ends with a focus on a particular subset of the newly-found materials, 2D insulators characterized by very large band gaps. “They stood out from the rest of the group, and they could be useful for nanoscale transistors, where it is not easy to find suitable insulator materials because of the extremely small dimensions involved” Campi explains.
The group verified that some of these materials, in particular B2F8Na, F2H6O2 and B2O9S2 do indeed have most of the requirements for such an application, in terms of bandgap, dielectric constant, and effective masses. But more studies will be required to confirm the feasibility of using them for these applications.
The study could have several developments in the future, says Campi. “One would be to use a similar technique for one-dimensional materials, chains of atoms that are weakly bound to the rest of the structure. Another one would be to play with the 2D materials that we have found, for example by placing one layer of a material over one layer of another, to create structures that combine different properties for targeted applications”. This could be particularly interesting for photovoltaics or photolytic water splitting, where a tailored disposition of electron-hole pairs is key.
In the long run, Campi says, the current database could be expanded to also include speculative materials, that do not have an experimental 3D counterpart but are similar to existing structures. “That is a bit more ambitious, but at least in some cases it could guide experimentalist to try and synthesize interesting new materials”.
Reference
D. Campi, N. Mounet, M. Gibertini, G. Pizzi, N. Marzari, "Expansion of the Materials Cloud 2D Database", ACS Nano 17, 12, 11268–11278 (2023). DOI: https://doi.org/10.1021/acsnano.2c11510
Low-volume newsletters, targeted to the scientific and industrial communities.
Subscribe to our newsletter